|
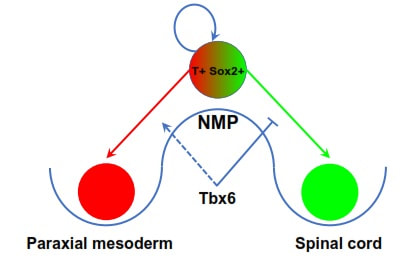
by Alok Javali A normal adult human body is made up of few trillion cells. These cells are classified into more than two hundred different types. Cells making blood are clearly very different from the ones that make bones, muscles or the brain, in appearance as well as function. This spectacular cell type diversity in the adult individual, originates from a single cell, that is the fertilized egg. How does a single cell divide and specialize into so many different types? This is a central question in the field of developmental biology. At the beginning, a fertilized egg undergoes a series of cell divisions to form the embryo. As the embryo develops further, various cell types emerge as a result of a series of choices made by the embryonic cells. In an individual organism, all the cells have identical DNA molecules in the nucleus, called the genome. The genome acts as a complex ‘encrypted map’ to guide cells in their developmental path. The fate of the cells mainly depends on the way the genome is decrypted. The cells employ various decrypting ‘tools’ referred to as regulatory elements, such as transcription factors, growth factors, micro RNAs and epigenetic regulators etc. A distinct set of decrypting tools are ‘switched on’ in the cells since they experience varied external cues at different places and times in the embryo. This allows interpretation of the genome in distinct ways leading the cells to choose different developmental paths and thus, eventually become different cell types. Studying the decrypting tools used by specific cells is an important endeavour, because it helps us understand how cells make choices and how things go wrong during developmental defects. A recent study in our laboratory has shown how some of these tools control the fate of a stem cell population, which gives rise to spinal cord as well as muscles and skeleton below the neck. During embryonic development, the elongation of the body below neck along the head to tail axis is executed by ‘axial progenitor cells’. These cells are located near the tail-end of the embryo. A major subgroup of these cells is the group of stem cells known as the neuromesodermal progenitors (NMPs). These cells are key to growth of the body lengthwise, in mammals, birds and reptiles. Discovery of the NMPs has dramatically changed our understanding of how the body plan is setup during the development of these animals. In a growing embryo of these organisms, NMPs choose one of the three developmental routes. They form a part of the spinal cord, a part of the muscle/skeleton forming tissue called somites or they simply self-renew (divide to make more copies) to form a greater number of NMPs. The balanced contribution of NMPs to somites and developing spinal cord results in elongation of the embryo, whereas, the self-renewing property of NMPs ensures the sustenance of elongation by continuously providing NMPs. NMPs are equipped with the appropriate level of ‘tools’ required for them to form either part of spinal cord or somites. Research conducted in our lab has identified a ‘molecular switch’ that guides NMPs in the direction of formation of somites. NMPs are characterized by simultaneous presence of key molecular tools which are required for formation of somites as well as spinal cord. An ongoing study in our lab found the additional presence of a pro-somite protein Tbx6, precisely in a subset of NMPs that are on the verge of taking the route to become somites. This opened up a possibility that Tbx6 acts as molecular switch in NMPs, causing cells to choose to become somites rather than spinal cord. To verify this, we observed the mouse embryos lacking Tbx6. Just as predicted, all the NMP derived somite forming cells were transformed into extra spinal cords in the absence of Tbx6. Additionally, previous studies from a different research group suggest that Tbx6 functions by inhibition of a key pro-spinal cord factor. In combination, these observations confirm that Tbx6 acts as a ‘genetic switch’ in NMPs that shuts the path that leads to the spinal cord formation, thus, allowing these cells to form somites. Tbx6 belongs to a class of proteins called transcription factors which binds to specific parts of the genome to decrypt it. We are currently interpreting the mechanism of genome decryption by Tbx6, in our lab. Further observations in the embryos lacking Tbx6, also revealed that, these embryos with extra spinal cord tissues in the place of somites, failed to elongate in normal manner. The somites to be developed between forelimb and hindlimb (equivalent of human hands and legs respectively) were transformed into two additional spinal cord like tissues. However, beyond the hindlimb, towards the tail of the embryo, somites were transformed into four additional spinal cord tissues. The effect of removal of Tbx6 was more severe towards the tip of the tail where all the cells were completely transformed into spinal cord progenitor cells forming a bulge of cells instead of a normal pointy tip. This bulge marked cessation of the elongation of the embryo. These observations suggest that the precise balance between contribution of NMPs to the developing spinal cord as well as the somites is critical for the normal elongation of the embryos. This suggests that development of NMPs may have something to do with regulation of body size. Therefore, following NMP development in different organisms across mammals, birds and reptiles may also help us explain the enormous diversity in size present in this group of animals. Understanding developmental events has important applications in the field of regenerative medicine. The knowledge generated with these studies can be harnessed to grow clinically important cell types in labs. For example, lab grown NMPs from embryonic stem cells have already been used to model the patterning of spinal cord by a research group. A recent study by another group has derived another clinically important cell type called neural crest cells from lab grown NMPs. Currently, lab grown tissues and organs are already being used to test drugs on them and model diseases. A detailed understanding of developmental process further generates hope for using lab grown cells to repair or regenerate damaged tissues, for example, to use such lab grown NMPs in treating spinal cord injuries or any related diseases. In conclusion, studying embryonic development can provide us new knowledge about the natural world as well as help generate novel avenues in the field of medicine. The study described here was published in the journal Development. The team involved in the study included Alok Javali, Aritra Msra, Karolis Leonavicius, Debalina Acharya, Bhakti Vyas and led by Dr. Ramkumar Sambasivan from the Institute for Stem Cell Biology and Regenerative Medicine, Bengaluru. The research article can be accessed at http://dev.biologists.org/content/144/24/4522.long
0 Comments
Leave a Reply. |
ArchivesCategories |

 RSS Feed
RSS Feed
